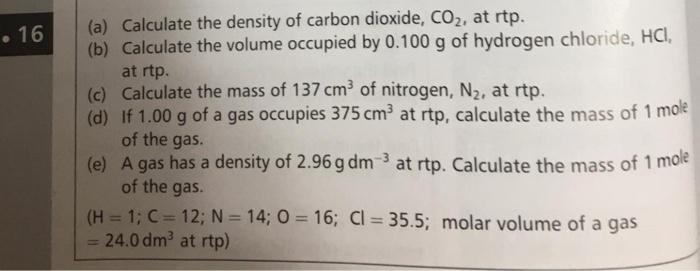SOLVED: 1.. Determine the mass of 0.4mol of zinc hydroxide (Zn (OH)) How many moles are in 8.28g of potassium carbonate (KCOs)? How many carbon dioxide molecules are in 11.0 g of
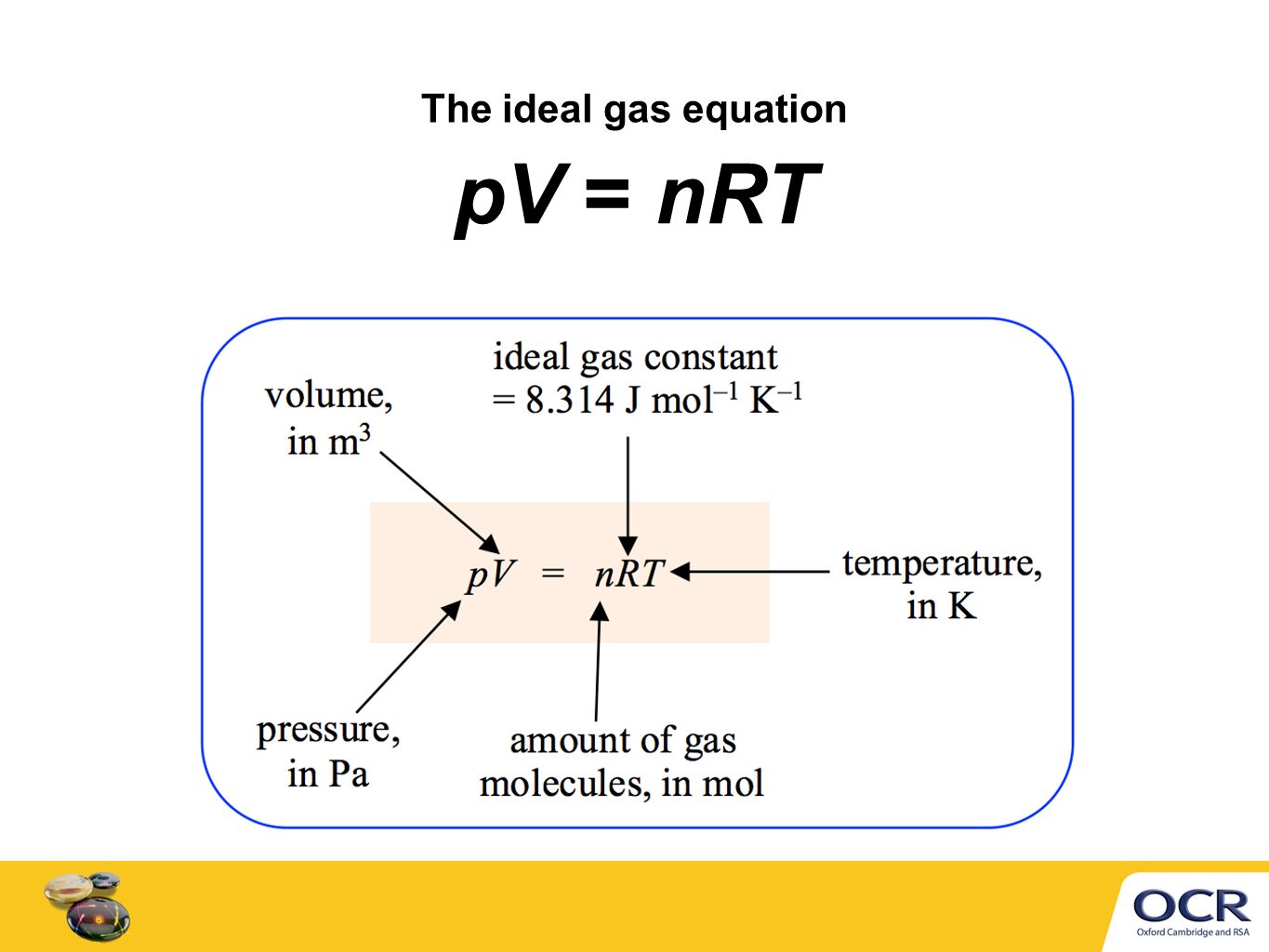
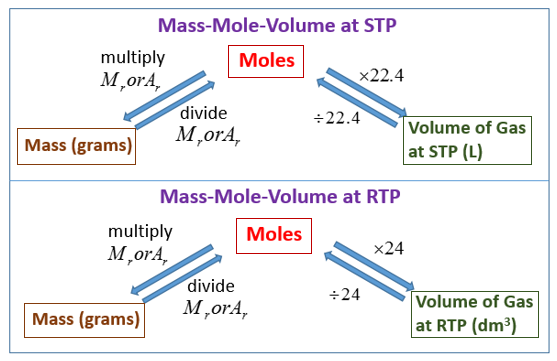
The ideal gas equation. Room temperature and pressure, RTP Limitations At RTP, 1 mol of gas molecules occupies 24.0 dm 3 Conditions are not always room. - ppt download

The ideal gas equation. Room temperature and pressure, RTP Limitations At RTP, 1 mol of gas molecules occupies 24.0 dm 3 Conditions are not always room. - ppt download

The ideal gas equation. Room temperature and pressure, RTP Limitations At RTP, 1 mol of gas molecules occupies 24.0 dm 3 Conditions are not always room. - ppt download

Calculate the volume of carbon(IV)oxide at r.t.p produced when 5.0 g of calcium carbonate is strongly heated.(Ca=40.0, C= 12.0,O = 16.0,1 mole of gas =22.4...